At present, the form of prevention and control of the new coronavirus is still very severe. As the first step of nucleic acid detection, the importance of sample collection and preservation is undoubted. The inspection industry often says “garbage in, garbage out” is important. The first influencing factor is sample collection. If there is a problem with the sample collection, no matter how well the subsequent work is done, the result will be invalid. Choosing the correct virus preservation solution and collection tube can make the new crown detection more effective
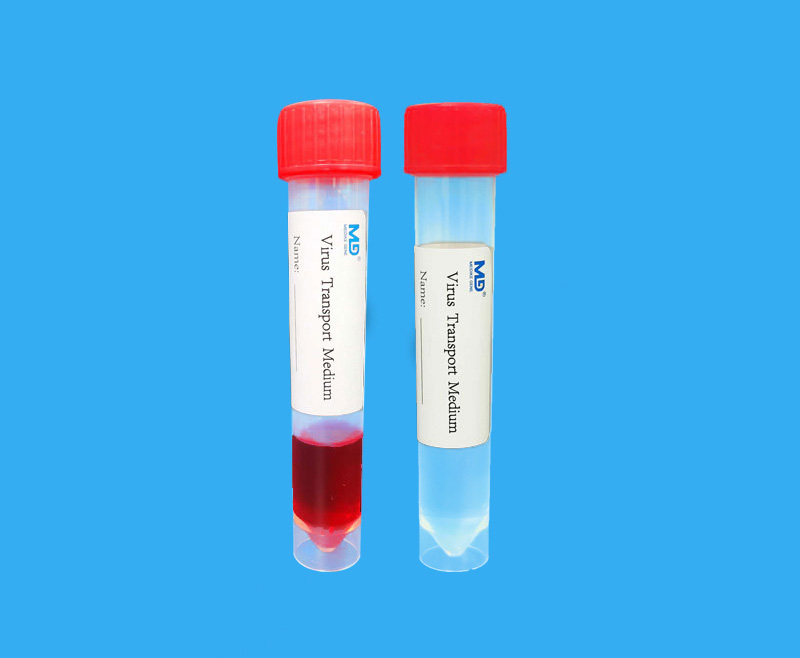
Almost all virus preservation liquids on the market directly preserve live viruses, causing medical staff in sampling, transportation and testing to face a higher risk of infection. In response to this situation, the front line of anti-epidemics is in need of samples that can directly inactivate the virus. Preservation solution, but it should be noted that while inactivating the virus, it should also consider whether the preservation solution can stably preserve the integrity of the viral nucleic acid, so as to avoid degradation of the nucleic acid in the sample before the test, resulting in a “false negative” nucleic acid test. Shenzhen Medico does not contain guanidine salt virus preservation solution to minimize the risk of “false negatives”.
We compared the effects of the preservation solution containing guanidine hydrochloride and guanidine isothiocyanate on the market with the Shenzhen Medico preservation solution without guanidine salt in the preservation of poultry coronavirus at 37°C. It turns out that the main ingredient is the preservation solution of guanidine salt, and the preservation effect is very unsatisfactory.
The same conditions are 37°C. After storage for 1-7 days, the preservation efficiency of viral nucleic acid is basically constant at 100% when using Shenzhen Medico preservation solution; while the preservation efficiency of viral nucleic acid is gradually reduced when the other two guanidine salt components are used. , By the 7th day, it was already less than 20%, indicating that the RNA is degrading violently over time
Guanidine salt (guanidine isothiocyanate or guanidine hydrochloride, etc.), as a classic protein denaturant, is often used for cell lysis in nucleic acid extraction, and can also achieve the effect of inactivating viruses, but the guanidine salt component does not have the ability to operate under normal temperature conditions. The ability to preserve viral nucleic acid is very easy to cause sample degradation, so it is not suitable for the preservation of new coronavirus samples, so it is strongly recommended that you choose a virus preservation solution that does not contain guanidine salts.
Here, we appeal to the majority of medical workers, if your sampling purpose is for nucleic acid testing and you do not need to cultivate live viruses, then you need to pay attention to the following aspects when choosing a virus preservation solution:
• Use a preservation solution that can inactivate the virus
The current nucleic acid detection is to detect the viral RNA in the sample, and the virus needs to be lysed. Therefore, as long as the integrity and stability of the viral nucleic acid can be ensured, there is no need to preserve the live virus. Therefore, in the current situation of fighting the new coronavirus epidemic, it is more ideal to inactivate the virus while collecting samples. Some manufacturers on the market have also introduced inactivated storage solutions, but the inactivated ingredients used are guanidine salts. The results of comparative experiments found that the guanidine salt component storage solutions do not have the ability to preserve viral nucleic acids under normal temperature conditions, which can easily cause sample degradation. It is not suitable for the preservation of new coronavirus samples. Therefore, we recommend a virus preservation solution containing non-guanidine salt protein denaturants, which can inactivate the virus and ensure that the viral nucleic acid is not degraded when stored at room temperature.
• The volume of the preservation solution in the sampling tube should be appropriate
It is very important to choose a storage solution with an appropriate volume: (1) If each sample is sampled separately, it is recommended that you choose a virus sampling tube with 1ml storage solution. Not only the sampling volume can meet the needs of downstream nucleic acid extraction and detection, but also compared with 3ml storage solution. The concentration of the virus has increased by 3 times! ②If a large-scale initial screening and mixed test is performed, the swab samples of 3-5 people are placed in the same sampling tube. It is recommended that you use a virus sampling tube with 3ml preservation solution to ensure that the sample volume can meet Downstream nucleic acid extraction needs, while improving detection efficiency and reducing detection costs.
• Use a virus preservation solution that can transport and preserve samples at room temperature
Because of the instability of RNA, traditional virus sampling tubes need to be transported to the laboratory within 48 hours at 4°C. If the transport temperature is not up to standard, viral nucleic acid may be degraded, resulting in “false negative” test results! In contrast If Medico uses a virus storage solution that can transport and stably store viral nucleic acids at room temperature, the problem of sample transportation can be solved, thereby improving the accuracy of the test results.
• Use a virus preservation solution that can protect samples from high temperature inactivation
The relevant guidelines of the National Health Commission require high temperature inactivation of the sample before extracting viral nucleic acid (treatment above 56°C for 30 minutes), but the Beijing Center for Disease Control and Prevention, Beijing Preventive Medicine Research Center and other institutions published research papers in Clinical Chemistry It shows that high temperature inactivation will cause the degradation of viral nucleic acid, thereby reducing the detection rate of viral nucleic acid, which is one of the reasons for the high false negative rate of new coronavirus nucleic acid detection. Experiments have verified that when using Medico virus sample preservation solution to store samples, high temperature inactivation has no significant effect on the integrity of coronavirus nucleic acid.
The virus preservation solutions developed and produced by Shenzhen Medico are divided into inactivated and non-inactivated types. Different preservation solutions can be selected according to different experimental requirements and testing conditions. If you have any questions or needs, please call to inquire. Choosing Shenzhen Medico Virus Preservation Solution is your right choice.
find it on: https://www.medicoswab.com/products/transport-medium/virus-transport-medium/




















